Leaders in the Field of Minimally Invasive Spine Surgery
NEWS: Dr. Neel Anand Appointed President of The Society for Minimally Invasive Spine Surgery (SMISS)
The Anand Spine Group is excited to announce that renowned spine surgeon Dr. Neel Anand, Professor of Orthopaedic Surgery and Neurosurgery and Medical Director of the Department of Orthopaedic Surgery and Neurosurgery at Cedars Sinai Medical Center at Cedars-Sinai Medical Center in Los Angeles, has been appointed President of The Society for Minimally Invasive Spine Surgery (SMISS) for the 2025–2026 term.
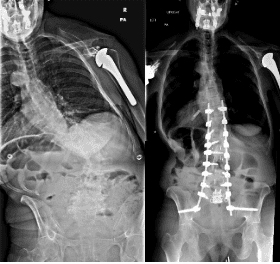
Correction of adult spinal deformity (ASD) through minimally invasive techniques is a challenging endeavor and has typically been reserved for experienced surgeons. This publication aims to be the first high-resolution technique guide to demonstrate a reproducible technique for ASD correction utilizing circumferential minimally invasive surgery (cMIS) without an osteotomy. The Segmental Interbody, Muscle-Preserving, Ligamentotaxis-Enabled Reduction (SIMPLER) technique is a novel ligamentotaxis-based scoliosis surgery that represents a paradigm shift from traditional osteotomies toward patient-specific correction.
The Anand Spine Group takes great pride in sharing the latest news in minimally invasive spine surgery. Dr. Anand has been on multiple medias over the years talking about latest technology and techniques in spine surgery. He is considered the expert in minimally invasive spine care and is leading the way in minimally invasive spine deformity correction. He loves to educate and provide the latest up to date technology that is available.
As a nationally and internationally renowned orthopedic spine surgeon and prolific researcher, Dr. Neel Anand is invited to present at the spine industry's most prestigious conferences and meetings. Stay up to date with Dr. Anand's latest talks here.
FREMISS
It is through the Foundation for Research and Education in Minimally Invasive Spine Surgery (FREMISS) that global aid is provided. The FREMISS Global Aid program is designed to benefit not only surgeons, but also patients in under privileged regions of the world. With your support, we can expand our research, strengthen our programs and improve the future of spinal care to patients around the world.
Additionally, FREMISS supports several educational opportunities for spine surgeons. The Comprehensive Minimally Invasive & Outpatient Spine Surgery Fellowship is an academic experience offered to spine surgeons who desire to train in minimally invasive techniques with Dr. Anand and conduct research. Upon graduation, fellows are encouraged to take their knowledge into various communities around the country. The Visiting Surgeon Program welcomes spine surgeons from around the world to spend time with Dr. Anand in clinic and in the operating room to better understand the philosophy and application of minimally invasive and outpatient techniques.
Thank you for your generosity which allows us to continue our mission.